E A Olsen, M Harries, A Tosti, W Bergfeld, U Blume-Peytavi... S Vano-Galvan. Guidelines for Clinical Trials of Frontal Fibrosing Alopecia: Consensus Recommendations from the International FFA Cooperative Group (IFFACG)
Br J Dermatol. 2021
"The publication of this article of consensus is an important step towards clinical trials on frontal fibrosing alopecia" - Dr. Sergio Vañó Galván -
Summary:
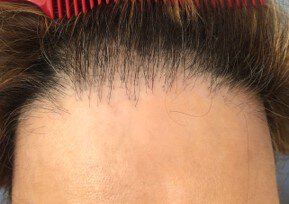
Background: Frontal fibrosing alopecia (FFA) has become one of the most common causes of cicatricial alopecia worldwide, but lacks clear aetiology and robust clinical trial evidence for efficacy and safety of agents currently used for treatment.
Objectives: To enable data to be collected worldwide on FFA using common criteria and assessment methods.
Methods: A multi-centre, international group of experts in hair loss was convened by email to create consensus recommendations for clinical trials. Consensus was defined at >90% agreement on each recommended part of these guidelines.
Results: Standardized diagnostic criteria, severity rating, staging, and investigator and patient assessment of scalp hair loss and other clinical features of FFA were created.
Conclusions: These guidelines should allow the collection of reliable aggregate data on FFA and further efforts in both clinical and basic research to close knowledge gaps in this condition.
Why do you highlight this publication?
This article reflects the consensus of an international research group on diagnostic criteria and monitoring methods for frontal fibrosing alopecia. This alopecia has a very negative impact on quality of life and is currently the most common scarring alopecia. The International Fibrosing Frontal Alopecia Working Group (IFFACG) was set up in 2018, bringing together more than 40 experts from five continents. The publication of this article is an important step towards clinical trials on this type of alopecia.
Publication commented by:
Dr. Sergio Vañó Galván
Dermatology Service of Hospital Universitario Ramón y Cajal