Rodríguez-Beltrán J, Sørum V, Toll-Riera M, de la Vega C, Peña-Miller R, San Millán Á. Genetic dominance governs the evolution and spread of mobile genetic elements in bacteria.
Proc Natl Acad Sci U S A. 2020
"Genetic dominance dictates the evolution and dissemination of antibiotic resistance in bacteria" - Dres. Jerónimo Rodríguez-Beltrán y Álvaro San Millán -
Summary:
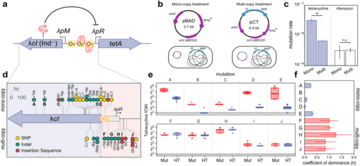
Mobile genetic elements (MGEs), such as plasmids, promote bacterial evolution through horizontal gene transfer (HGT). However, the rules governing the repertoire of traits encoded on MGEs remain unclear. In this study, we uncovered the central role of genetic dominance shaping genetic cargo in MGEs, using antibiotic resistance as a model system. MGEs are typically present in more than one copy per host bacterium, and as a consequence, genetic dominance favors the fixation of dominant mutations over recessive ones. In addition, genetic dominance also determines the phenotypic effects of horizontally acquired MGE-encoded genes, silencing recessive alleles if the recipient bacterium already carries a wild-type copy of the gene. The combination of these two effects governs the catalog of genes encoded on MGEs. Our results help to understand how MGEs evolve and spread, uncovering the neglected influence of genetic dominance on bacterial evolution. Moreover, our findings offer a framework to forecast the spread and evolvability of MGE-encoded genes, which encode traits of key human interest, such as virulence or antibiotic resistance.
Why do you highligth this publication?
Antibiotic resistance is one of the major threats facing public health. Bacteria usually become resistant to antibiotics by the horizontal acquisition of mobile genetic elements (MGE). In this context, it is important to develop new frameworks to predict which antibiotic resistance genes are likely to spread among bacterial populations. In this work, we discovered that genetic dominance determines which genes are likely to be present on mobile genetic elements. Our results offer a new understanding of the forces that shape the genetic repertoire of MGE, and help to predict which antibiotic resistance genes are likely to spread horizontally, which will be useful to rationally assess the risk of spread for resistance.
Publication commented by:
Dres. Jerónimo Rodríguez-Beltrán y Álvaro San Millán
Plasmid Biology and Evolution Lab
Biology and Evolution of Microorganisms Group
IRYCIS