Cevallos K, Abad JL, Babiy B, Sacristán S, Hidalgo A, Senra Y, Busto R, Martínez J, Abad D, Bujons J, Fábrias G, Pastor Ó. Synthesis and Biological Evaluation of a New Dihydroceramide Desaturase Inhibitor for the Treatment of MASLD
J Med Chem. 2026
"This is a promising proof-of-concept showing that blocking lyphotoxic ceramides with small molecule inhibitors of DEGS1 can be beneficial in MASLD". - Dr Óscar Pastor Rojo
Summary:
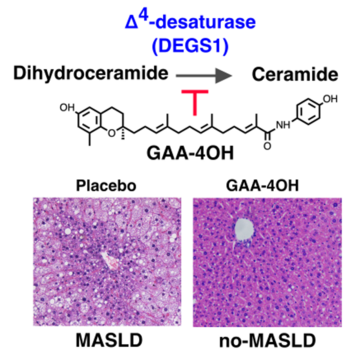
Ceramides and dihydroceramides are associated with obesity, type 2 diabetes, and metabolic dysfunction-associated steatotic liver disease (MASLD). Despite their pathogenic relevance, effective strategies to reduce ceramide levels remain lacking. Delta-tocotrienol (δ-TT) derivatives have been reported as weak inhibitors of dihydroceramide desaturase-1 (DEGS1), a key enzyme in ceramide biosynthesis. Here, we screened a library of δ-TT derivatives and identified GAA-4OH as a potent and irreversible inhibitor of DEGS1. In vitro, GAA-4OH exhibited nanomolar activity, surpassing existing compounds such as fenretinide. Kinetic assays and molecular docking simulations suggest that GAA-4OH may undergo oxidation to form a reactive iminoquinone that covalently blocks the enzyme catalytic cavity. In vivo, GAA-4OH administration in a mouse model of MASLD reduced ceramide-to-dihydroceramide ratios and improved steatosis, inflammation, and fibrosis. These benefits occurred without body weight loss and were correlated with reduced pro-inflammatory and pro-fibrogenic gene expression, without signs of toxicity, supporting its safety and potential as a therapeutic for MASLD.
Why do you highligth this publication?
This publication represents a step forward in the search for effective treatments for metabolic associated-dysfunction steatotic liver disease (MASLD), a highly prevalent condition that currently lacks effective and safe pharmacological options to address its underlying lipotoxic mechanisms. Our results show that GAA-4OH, a novel δ-tocotrienol-derived irreversible inhibitor of DEGS1, an enzyme involved in lipotoxic ceramide synthesis, reduce fat accumulation, the inflammatory response and fibrogenesis in mice. This is a promising proof-of-concept stablishing that selective DEGS1 inhibition without retinoid metabolism interference can be therapeutically beneficial in MASLD. GAA-4OH is positioned as a viable lead compound for further drug development in cardiometabolic diseases, with a patent already filed (EP 26 382 402.1).
Publication commented by:
Dr Óscar Pastor Rojo
LIPID METABOLISM & LIPIDOMICS AND PROTEOMICS CHARACTERIZATION (UCA-CLP). IRYCIS