Santos HDA, Zabala Gutiérrez I, Shen Y, et al. Rubio J, Jaque D. Ultrafast photochemistry produces superbright short-wave infrared dots for low-dose in vivo imaging.
Nat Commun. 2020.
“Published results are the first step towards establishing the nanoparticle-assisted infrared fluorescence imaging in pre-clinical research units” - Dr. Daniel Jaque -
Summary:
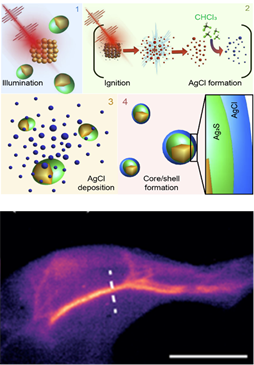
Optical probes operating in the second near-infrared window (NIR-II, 1,000-1,700 nm), where tissues are highly transparent, have expanded the applicability of fluorescence in the biomedical field. NIR-II fluorescence enables deep-tissue imaging with micrometric resolution in animal models, but is limited by the low brightness of NIR-II probes, which prevents imaging at low excitation intensities and fluorophore concentrations. Here, we present a new generation of probes (Ag2S superdots) derived from chemically synthesized Ag2S dots, on which a protective shell is grown by femtosecond laser irradiation. This shell reduces the structural defects, causing an 80-fold enhancement of the quantum yield. PEGylated Ag2S superdots enable deep-tissue in vivo imaging at low excitation intensities (<10mWcm−2) and doses (<0.5 mg kg−1), emerging as unrivaled contrast agents for NIR-II preclinical bioimaging. These results establish an approach for developing superbright NIR-II contrast agents based on the synergy between chemical synthesis and ultrafast laser processing.
Why do you highligth this publication?
Although there have been numerous occasions that nanotechnology can revolutionize pre-clinical research by developing new probes and contrast elements of dimensions below 100 nm. Nanotechnology is playing a particularly important role in the development of infrared luminescent probes for use in live image without the need for ionizing radiation or high-cost equipment (such as magnetic resonance imaging). However, infrared fluorescence in vivo imaging is not a popular technique in pre-clinical animal experimentation laboratories due to the lack of sufficiently bright and effective probes. In this work researchers have shown that it is possible to manufacture super-bright infrared fluorescent nanoparticles by using ultra-fast lasers innovatively. These super-nanoparticles have been used for high-resolution in vivo imaging and contrast of the cardiovascular system by administering ultra-low doses of super-nanoparticles as well as low excitation intensities. Published results are the first step towards establishing the nanoparticle-assisted infrared fluorescence imaging in pre-clinical research units.
Publication commented by:
Dr. Daniel Jaque
Department of Materials Physics
Universidad Autónoma de Madrid
Nanobiology group – IRYCIS