Zaragoza C. When RNAs come full circle: new regulators of healing after myocardial infarction
Eur Heart J. 2026
"Closing the loop: RNAs as New Orchestators of Healing after Myocardial Infarction" Dr. Carlos Zaragoza”
Summary:
Myocardial infarction (MI) continues to be a leading cause of heart failure worldwide, despite major advances in reperfusion strategies and chronic medical therapy. Beyond the initial ischemic insult, the way the injured myocardium heals mainly influences long-term outcome. Cardiac macrophages are central orchestrators of this response; however, the molecular switches that tune macrophage behavior after MI are only beginning to be understood.
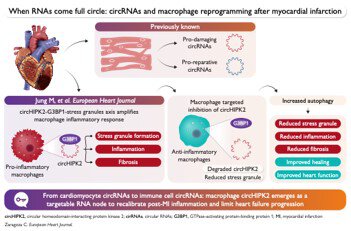
Circular RNAs (circRNAs) have emerged as an additional layer of gene regulation in cardiovascular biology. Their covalently closed-loop structure confers unusual stability, and they can interact with microRNAs, RNA-binding proteins, and components of the translational machinery. Over the last decade, animal models of myocardial infarction have revealed a remarkably complex circRNA atlas with opposing effects on injury and repair.
circHIPK2 is upregulated in inflammatory cardiac macrophages after infarction, and its temporal pattern closely tracks the inflammatory phase of post-MI remodeling. This editorial emphasizes recent research that examines the role of this lncRNA, focusing not on cardiac myocytes but on the immune system. It shows that circHIPK2 is not just a bystander; instead, it plays an active role in influencing macrophage phenotypes during cardiac healing.
Why do you highlight this publication?
Targeting circHIPK2 specifically in macrophages offers a promising approach to modulate inflammation after infarction, reduce fibrosis, and safeguard heart function. While realizing this therapeutic potential demands careful consideration of timing, delivery methods, and safety, the concept is compelling: by focusing on an RNA target within immune cells rather than just cardiomyocytes, we could steer ventricular healing towards a more favorable outcome. If successful, such strategies might revolutionize how we prevent and treat heart failure post-MI, shifting from a solely myocyte-focused perspective to one that fully embraces the role of immune cells in cardiac repair.
Publication commented by:
Dr. Carlos Zaragoza
CARDIOVASCULAR DISEASES. IRYCIS